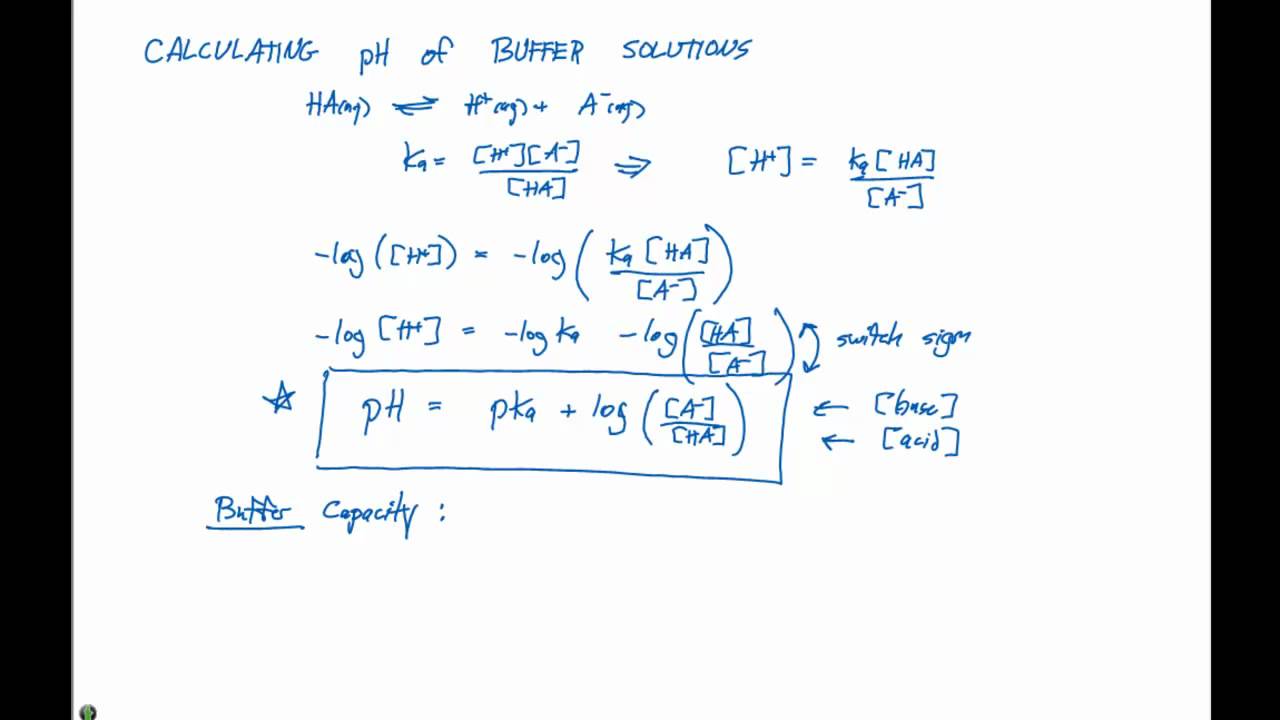
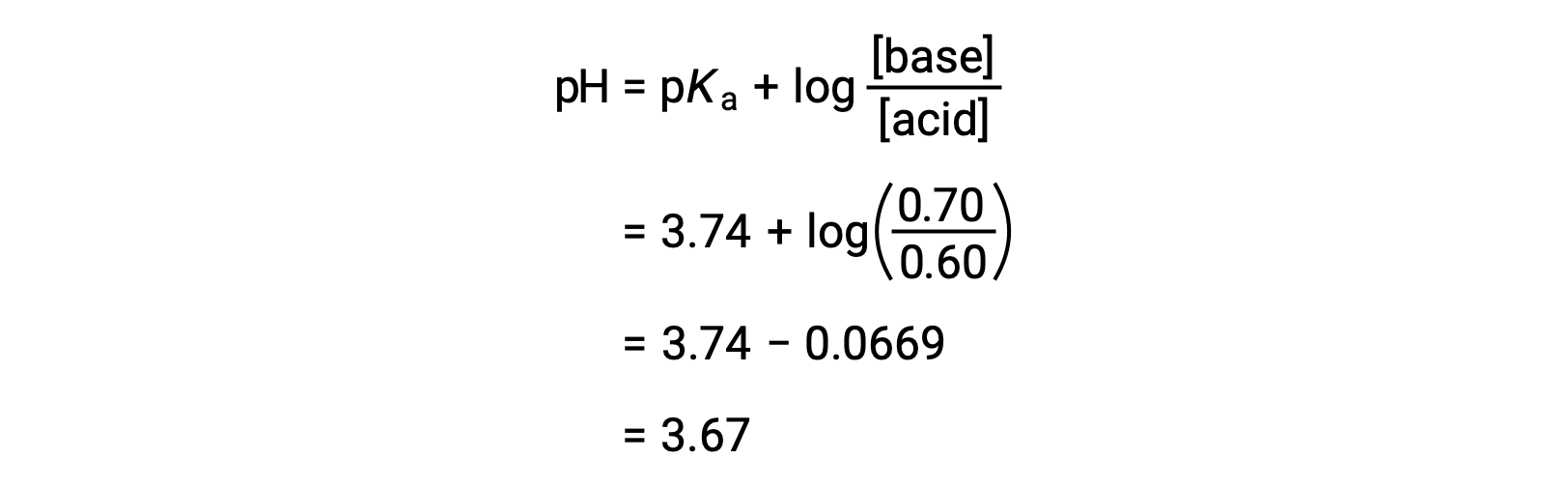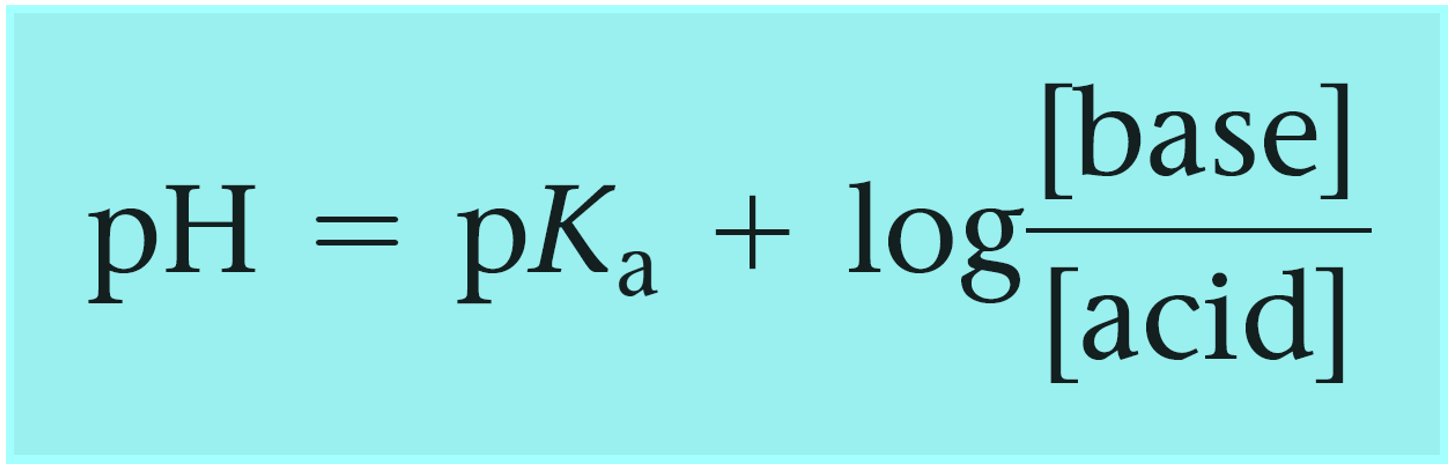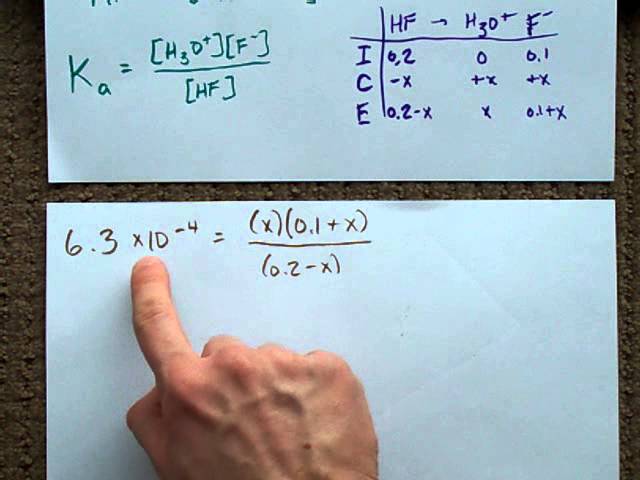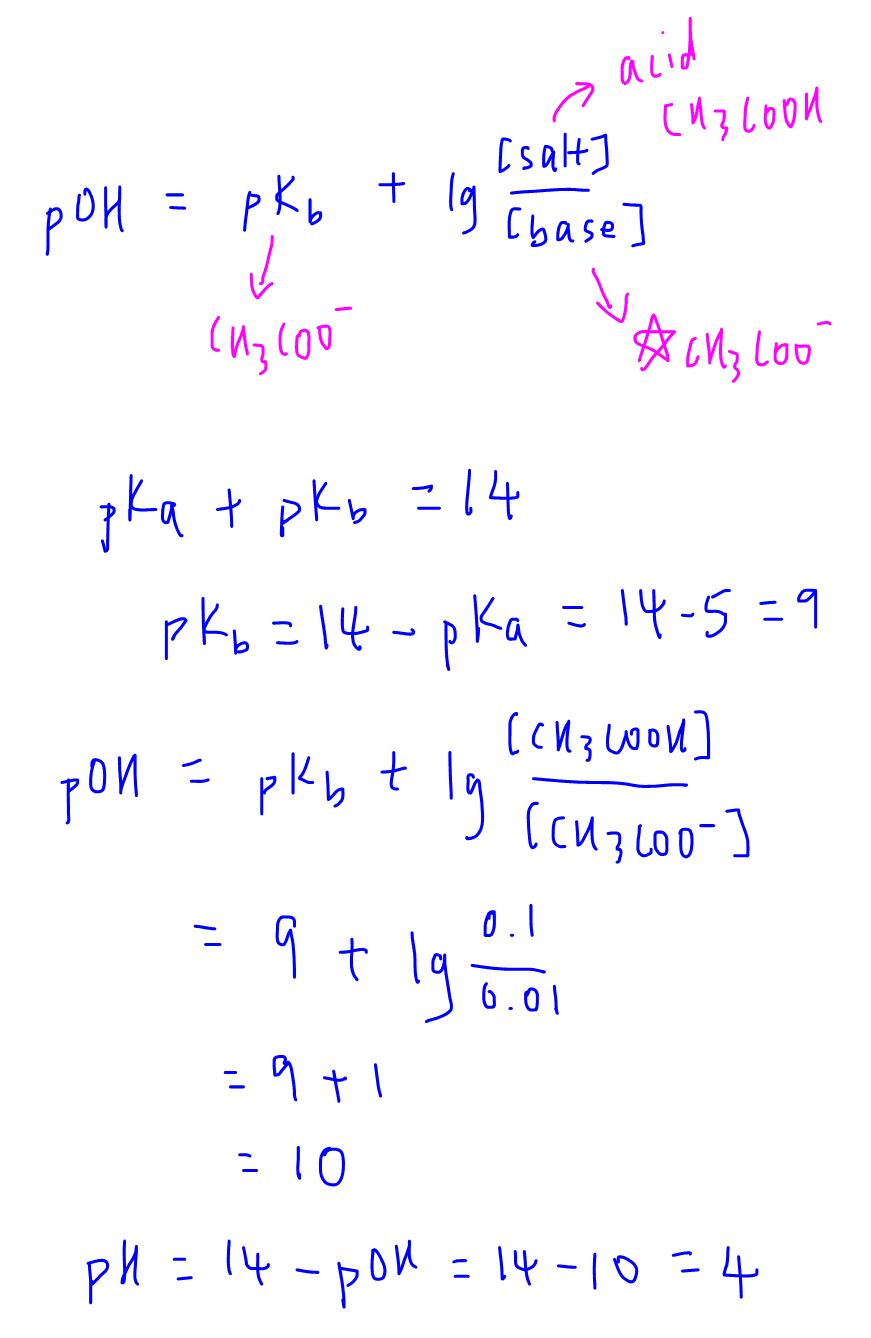SOLVED: Calculate the pH of a buffer solution made from 0.30 M hydrofluoric acid and 0.70 M sodium fluoride after the addition of 0.080 mol ofNaOHto ofthis solution Assume change in volume

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate.

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH and buffer capacity of a carbonic acid/ bicarbonate buffer solution.