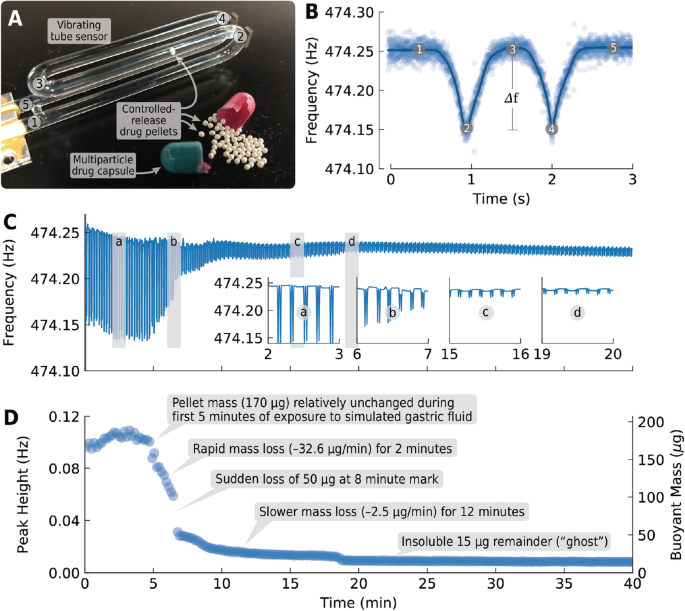
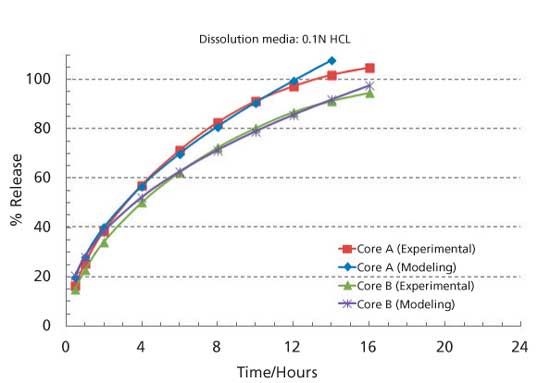
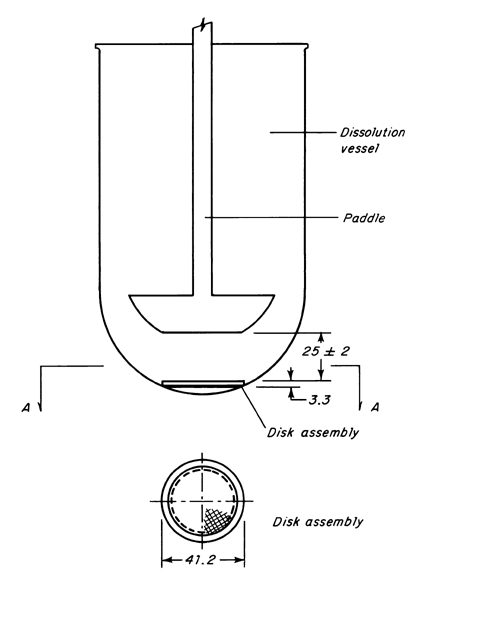
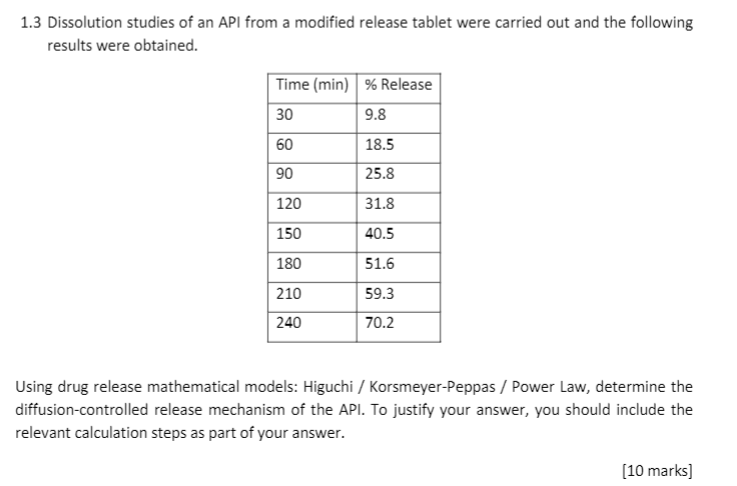
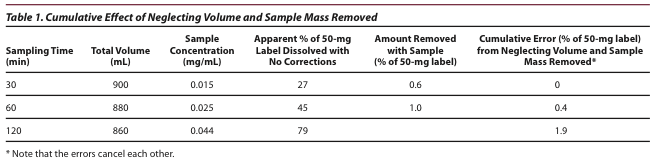
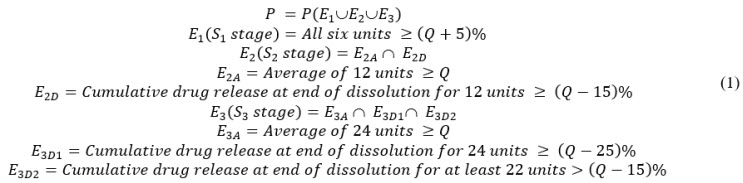A New Method for Evaluating Actual Drug Release Kinetics of Nanoparticles inside Dialysis Devices via Numerical Deconvolution - ScienceDirect

In Vitro–In Vivo Correlation in Cocrystal Dissolution: Consideration of Drug Release Profiles Based on Coformer Dissolution and Absorption Behavior | Molecular Pharmaceutics
![PDF] In-Vitro Dissolution Study and Shelf Life Calculation of Developed Sol-To-Gel Ocular Drug Delivery System of Brimonidine for Conjunctivitis during Accelerated Stability Study | Semantic Scholar PDF] In-Vitro Dissolution Study and Shelf Life Calculation of Developed Sol-To-Gel Ocular Drug Delivery System of Brimonidine for Conjunctivitis during Accelerated Stability Study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/095f28a3e1119f309d356c3494689d68e9a0cebf/3-Table3-1.png)
PDF] In-Vitro Dissolution Study and Shelf Life Calculation of Developed Sol-To-Gel Ocular Drug Delivery System of Brimonidine for Conjunctivitis during Accelerated Stability Study | Semantic Scholar

Biopharmacotechnical and physical properties of solid pharmaceutical forms containing rutin commercially acquired in Juiz de Fora city, Brazil
Comparative statistical analysis of the release kinetics models for nanoprecipitated drug delivery systems based on poly(lactic-co-glycolic acid) | PLOS ONE
![PDF] In-Vitro Dissolution Study and Shelf Life Calculation of Developed Sol-To-Gel Ocular Drug Delivery System of Brimonidine for Conjunctivitis during Accelerated Stability Study | Semantic Scholar PDF] In-Vitro Dissolution Study and Shelf Life Calculation of Developed Sol-To-Gel Ocular Drug Delivery System of Brimonidine for Conjunctivitis during Accelerated Stability Study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/095f28a3e1119f309d356c3494689d68e9a0cebf/3-Table5-1.png)
PDF] In-Vitro Dissolution Study and Shelf Life Calculation of Developed Sol-To-Gel Ocular Drug Delivery System of Brimonidine for Conjunctivitis during Accelerated Stability Study | Semantic Scholar
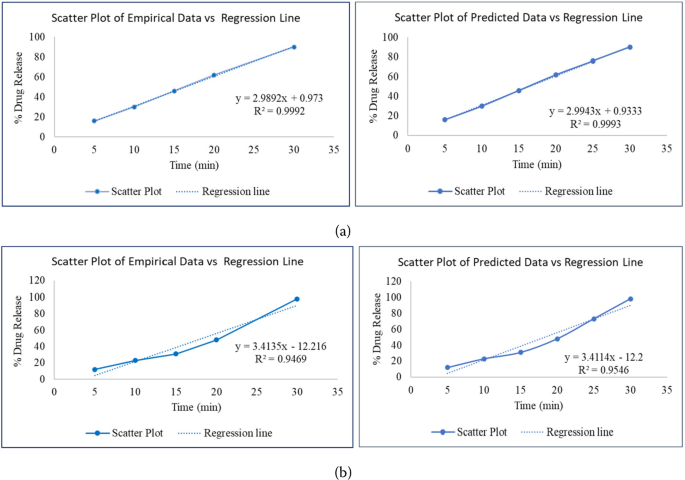
Optimizing similarity factor of in vitro drug release profile for development of early stage formulation of drug using linear regression model | Journal of Mathematics in Industry | Full Text
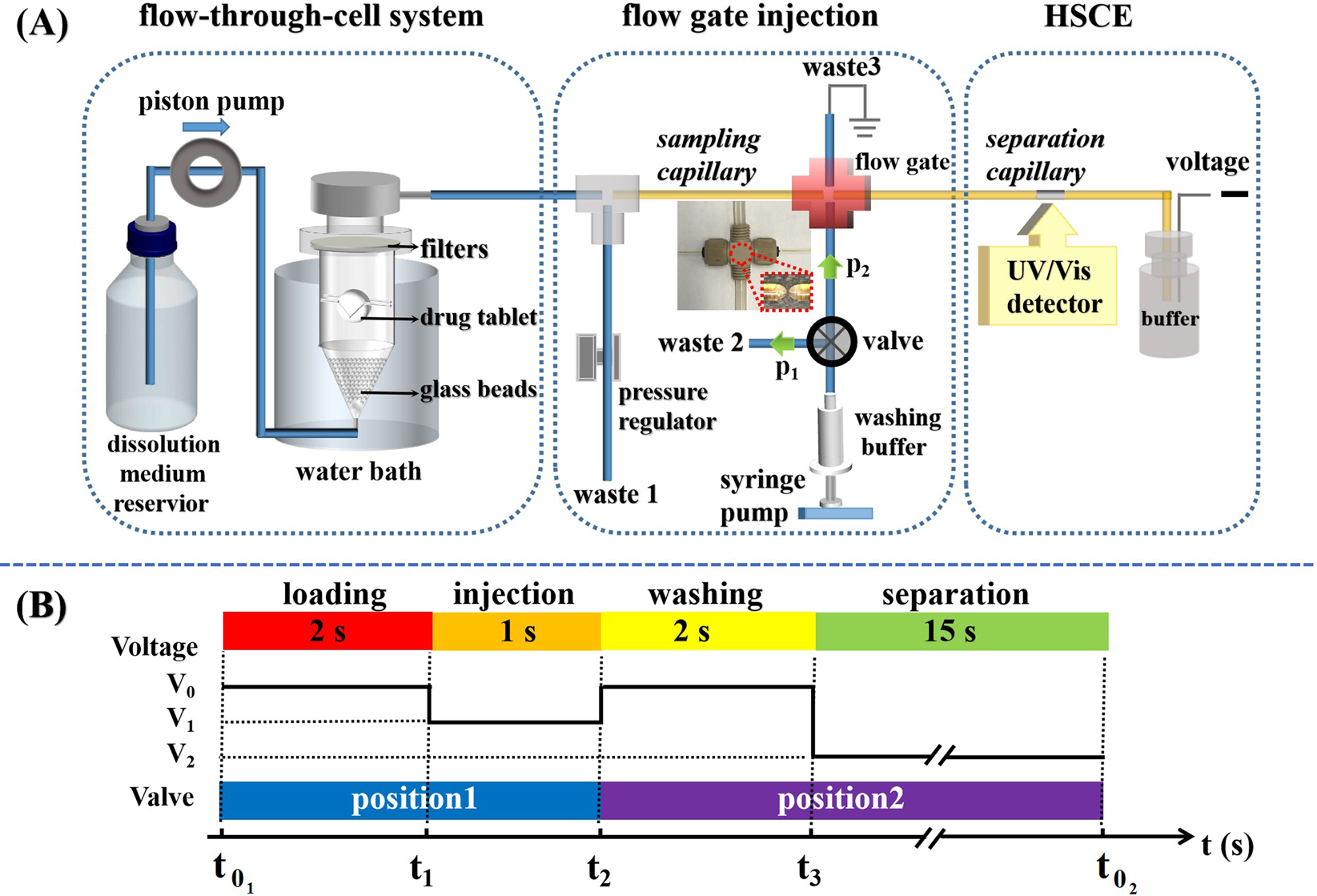
Automatic Dissolution Testing with High-Temporal Resolution for Both Immediate-Release and Fixed-Combination Drug Tablets | Scientific Reports
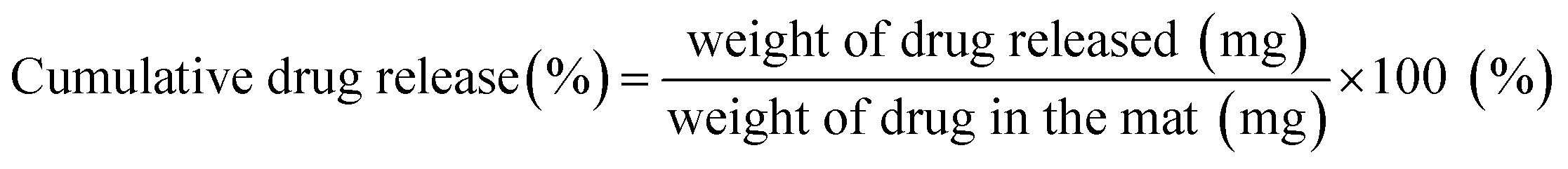
Early stage release control of an anticancer drug by drug-polymer miscibility in a hydrophobic fiber-based drug delivery system - RSC Advances (RSC Publishing) DOI:10.1039/C8RA01467A