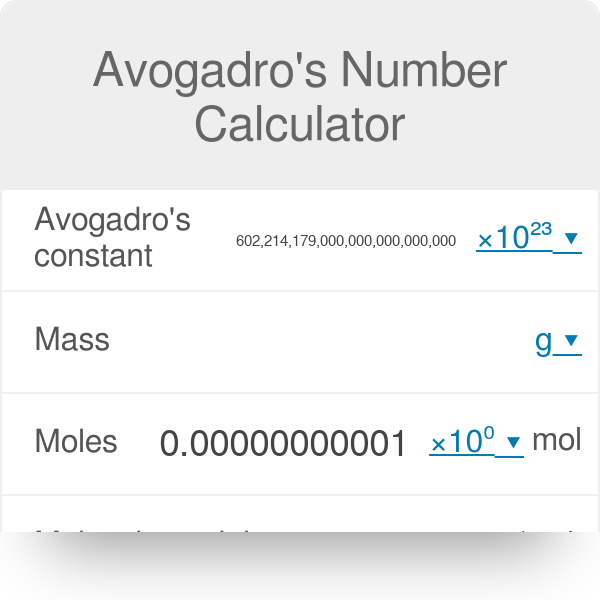
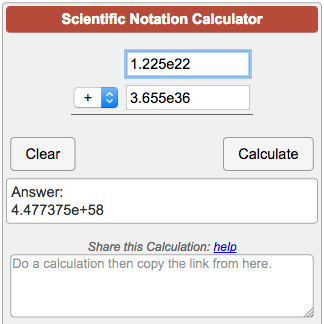
I need help with my calculator why is it showing this answer i dont know what i did wrong tho Im using a casio fx-991cw : r/calculators
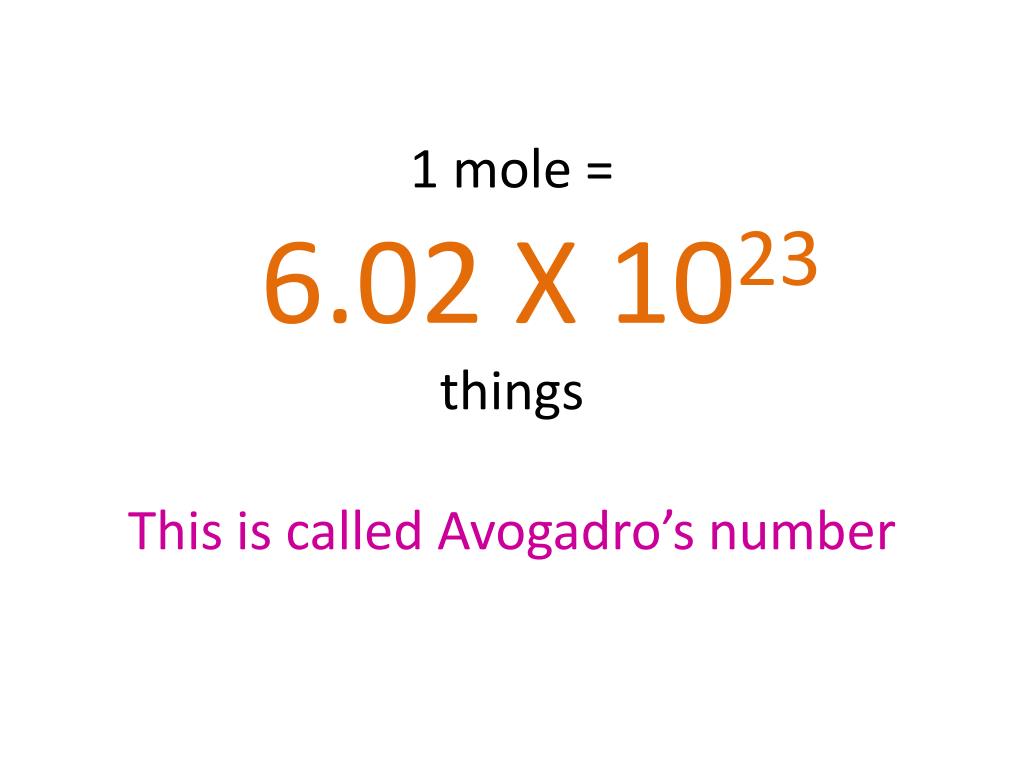
PPT - 1 mole = 6.02 X 10 23 things This is called Avogadro's number PowerPoint Presentation - ID:4272623

The Mole Notes. In every industry, there is a convenient way to measure a quantity of materials. For bakers, a baker's dozen = 13 For egg farmers, a dozen. - ppt download

Lithium has a BCC structure. Its density is 530 kg m^-3 and its atomic mass is 6.94 g mol^-1 . Calculate the edge length of a unit cell of Lithium metal: ( NA = 6.02 × 10^23 mol^-1 )

12 g C - 12 contains 6.022 × 10^23 atoms of carbon.(a) 6.022 × 10^23 is known as .............(b) Calculate the number of carbon atoms present in 48 g C - 12.(c)

The Mole Concept. What is a mole? IIn chemistry, a mole is a counting unit. Abbreviated mol. 11 mol = 6.022x10 23 representative particles. Avogadro's. - ppt download